Life Sciences
With the rising global health and access problems, the Life Sciences industry is focusing on the expansion of digital health, faster time to market, and cost-effective yet efficient solutions.
Total World Automation LLC has a competent workforce to support the industry’s strategic expansion projects across the board:
- API (active pharmaceutical ingredient)
- Formulation
- Biologics
While our team will deliver engineering and systems integration services for the execution of these projects, a remarkable emphasis will be paid to the digital transformation strategy with the goal of improving process yields and reducing process volume time cycles.

In practice, TWA will provide Safety in Design (SiD), Quality by Design (QbD), a comprehensive project life cycle assistance, computer system validation, and documentation. All of these in strict adherence to the following standards and regulations: CFR 21 Part 11, EU Annex 11, and GAMP5.
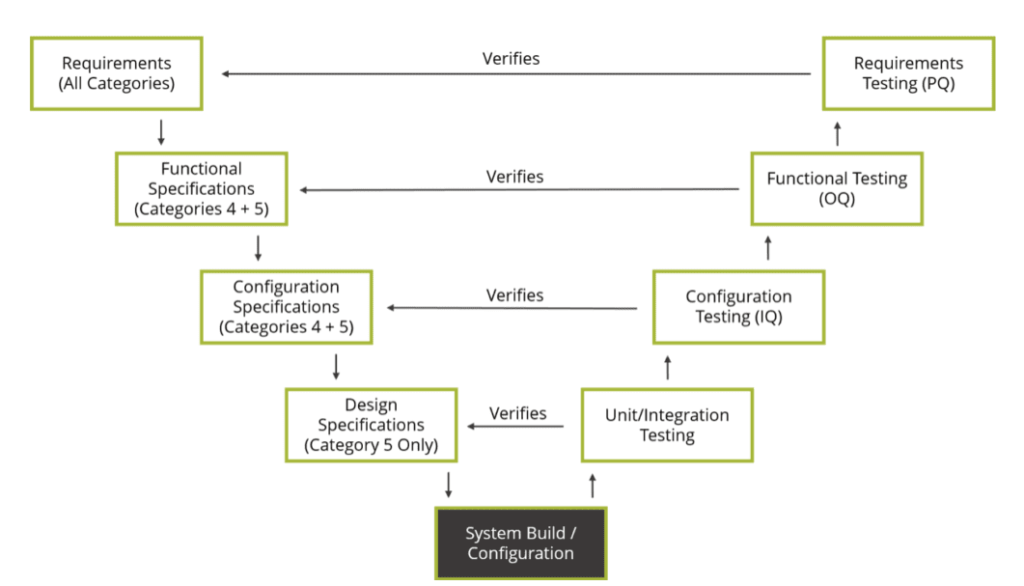
TWA enforces the “KARE” principle for all Life Sciences project requirements.
K – Kaizen Methodology
A – Agile Approach
R – Regulatory Compliance
E – Economical (yet efficient)
Come and collaborate with us!
We are as committed to meeting the regulatory compliant automation needs of your projects as you do for the health of your patients.
